DARUNAVIR MEVON FILM-COATED TABLETS 800 MG [SIN17329P]
Active ingredients: DARUNAVIR MEVON FILM-COATED TABLETS 800 MG
Product Info
DARUNAVIR MEVON FILM-COATED TABLETS 800 MG
[SIN17329P]
Product information
Active Ingredient and Strength | DARUNAVIR PROPYLENE GLYCOLATE 911.1 MG EQV DARUNAVIR - 800 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | PHARMATHEN INTERNATIONAL S.A - GREECE |
Registration Number | SIN17329P |
Licence Holder | NOVEM PHARMA PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J05A-E010 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Adult patients
Darunavir, in combination with 100 mg low dose ritonavir (Darunavir/rtv) and with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus (HIV) infection.
Pediatric patients
Darunavir, in combination with low dose ritonavir (Darunavir/rtv) and with other antiretroviral agents, is indicated for the treatment of HIV infection in treatment-experienced pediatric patients of 6 years and above and at least 20kg body weight.
In treatment-experienced adult and pediatric patients, the following points should be considered when initiating therapy with Darunavir/rtv:
Treatment history and, when available, genotypic or phenotypic testing should guide the use of Darunavir/rtv.
Dosing
4.2 Posology and method of administration
Darunavir must always be given with low dose ritonavir as a pharmacokinetic enhancer and in combination with other antiretroviral medicinal products. The prescribing information of ritonavir must therefore be consulted prior to initiation of therapy with Darunavir/rtv.
After therapy with Darunavir has been initiated, patients should be advised not to alter the dosage or discontinue therapy without instruction of their physician.
Method of administration: oral administration.
Darunavir must be taken with food. The type of food does not affect the exposure to Darunavir (see 5.2 Pharmacokinetic Properties – Absorption – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Dosage – Adults
Treatment-Naïve Adult Patients
The recommended oral dose of Darunavir tablets is 800 mg (one 800 mg tablet or two 400 mg tablets) taken with ritonavir 100 mg once daily and with food.
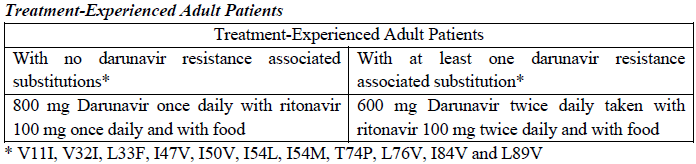
For antiretroviral treatment-experienced patients, HIV genotypic testing is recommended. However, when HIV genotypic testing is not feasible, the Darunavir/ritonavir 600/100 mg twice daily dosing is recommended.
The type of food does not affect the exposure to darunavir. Ritonavir (100 mg) is used as a pharmacokinetic enhancer of darunavir (see 4.5 Interactions and 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Dosage – Pediatrics
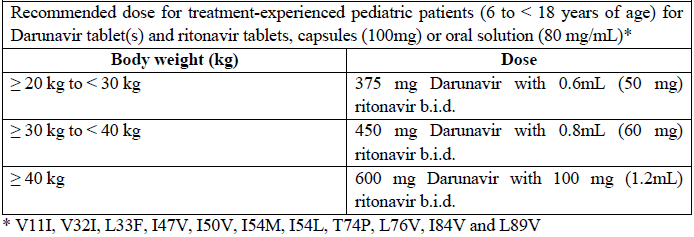
Antiretroviral treatment-experienced pediatric patients (6 to < 18 years of age)
The recommended dose of Darunavir/rtv for pediatric patients (6 to < 18 years of age and weighing at least 20 kg) is based on body weight (see table below) and should not exceed the recommended adult dose (600/100 mg b.i.d.). Darunavir tablets should be taken with ritonavir twice daily and with food. The type of food does not affect exposure to darunavir.
Antiretroviral treatment-experienced children less than 6 years of age and antiretroviral treatment-naïve pediatric patients
The safety and efficacy of Darunavir/ rtv in antiretroviral treatment-experienced children aged 3 to less than 6 years and in antiretroviral treatment-naïve pediatric patients have not been established.
Darunavir/rtv should not be used in children below 3 years of age (See 4.4 Warnings and precautions and 5.3 Preclinical safety data – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Pregnancy and postpartum
No dose adjustment is required for Darunavir/rtv during pregnancy and postpartum. Caution should be used in patients with concomitant medications which may further decrease darunavir exposure (see Section 5.1 Pharmacokinetic Properties - Special Populations - Pregnancy and Postpartum – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Missed dose(s)
If using the once daily regimen: in case a dose of Darunavir and/or ritonavir was missed within 12 hours of the time it is usually taken, patients should be instructed to take the prescribed dose of Darunavir and ritonavir with food as soon as possible. If this was noticed later than 12 hours after the time it is usually taken, the missed dose should not be taken and the patient should resume the usual dosing schedule.
If using the twice daily regimen: in case a dose of Darunavir and/or ritonavir was missed within 6 hours of the time it is usually taken, patients should be instructed to take the prescribed dose of Darunavir and ritonavir with food as soon as possible. If this was noticed later than 6 hours of the time it is usually taken, the missed dose should not be taken and the patient should resume the usual dosing schedule.
Special populations
Elderly (65 years of age and older)
Limited information is available on the use of Darunavir in patients 65 and older. Therefore, Darunavir should be used with caution in this age group (see 4.4 Warnings and Precautions and 5.2 Pharmacokinetic Properties – Elderly – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No dose adjustment is required in patients with renal impairment (see 4.4 Warnings and Precautions and 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No dose adjustment is required in patients with mild or moderate hepatic impairment. There are no pharmacokinetic or safety data available for subjects with severe hepatic impairment, therefore, Darunavir/rtv must not be used in patients with severe hepatic impairment (see 4.4 Warnings and Precautions and 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administration
Method of administration: oral administration.
Darunavir must be taken with food. The type of food does not affect the exposure to Darunavir (see 5.2 Pharmacokinetic Properties – Absorption – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Contraindications
4.3 Contraindications
Hypersensitivity to darunavir or to any of the excipients.
Darunavir and ritonavir are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. Darunavir/ rtv should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Examples include alfuzosin, astemizole, cisapride, colchicine (in patients with renal and/or hepatic impairment), dapoxetine, dronedarone, elbasvir/grazoprevir, the ergot alkaloids (e.g., ergotamine, dihydroergotamine, ergonovine and methylergonovine), ivabradine, lomitapide, lovastatin, lurasidone, midazolam (oral), naloxegol, pimozide, ranolazine, sildenafil (when used for treatment of pulmonary arterial hypertension), simvastatin, terfenadine, ticagrelor, and triazolam (see 4.5 Interactions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Patients taking Darunavir should not use products containing potent CYP3A inducers such as rifampin or St. John’s wort because co-administration may result in reduced plasma concentrations of darunavir. This may result in loss of therapeutic effect and development of resistance.
