POSACONAZOLE BIOCON DELAYED RELEASE TABLETS 100 MG [SIN17340P]
Active ingredients: POSACONAZOLE BIOCON DELAYED RELEASE TABLETS 100 MG
Product Info
POSACONAZOLE BIOCON DELAYED RELEASE TABLETS 100 MG
[SIN17340P]
Product information
Active Ingredient and Strength | POSACONAZOLE - 100 MG |
Dosage Form | TABLET, DELAYED RELEASE |
Manufacturer and Country | BIOCON PHARMA LIMITED - INDIA |
Registration Number | SIN17340P |
Licence Holder | GOLDPLUS UNIVERSAL PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J02AC04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Posaconazole Biocon delayed release tablets 100 mg is indicated for prophylaxis of invasive Aspergillus and Candida infections, including both yeasts and molds, in patients, 13 years of age and older, who are at high risk of developing these infections, such as patients with prolonged neutropenia or hematopoietic stem cell transplant (HSCT) recipients.
Posaconazole Biocon delayed release tablets 100 mg is indicated for use in the treatment of the following fungal infections in patients 13 years of age or older:
Refractory Invasive Fungal infections/Intolerant Patients with IFI: Fusariosis, zygomycosis, cryptococcosis, coccidioidomycosis, chromoblastomycosis, and mycetoma in patients with disease refractory to other therapy, or patients who are intolerant of other therapy. Refractoriness is defined as progression of infection or failure to improve after a minimum of 7 days of prior therapeutic doses of effective antifungal therapy.
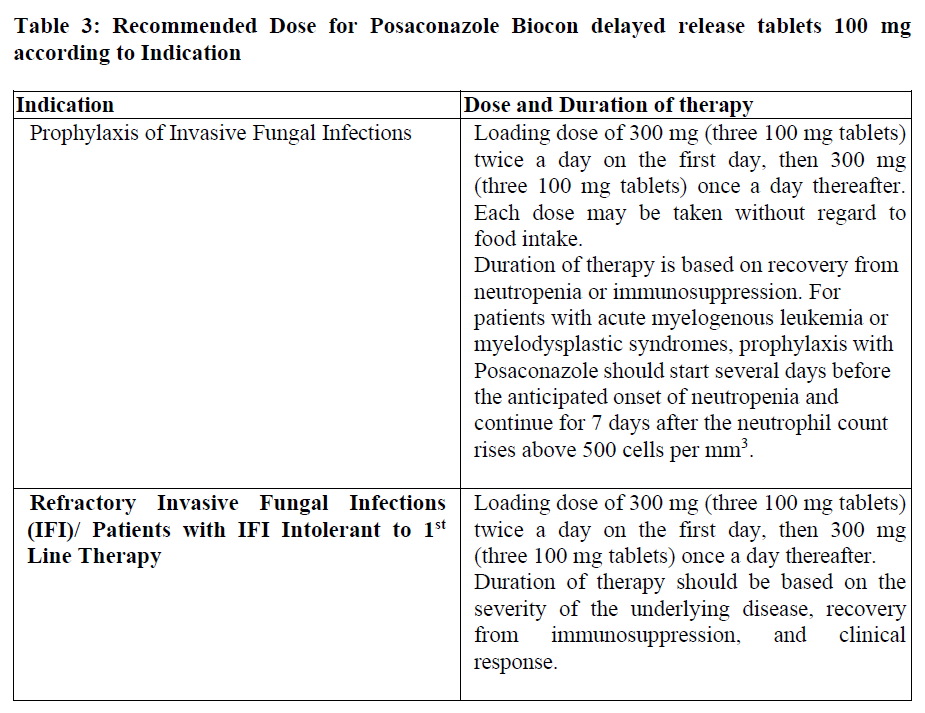
Dosing
4.2 Dosage and administration
Important Administration Instructions for Posaconazole Biocon delayed release tablets 100 mg
Non-Interchangeability between Posaconazole Biocon delayed release tablets 100 mg and Posaconazole Oral Suspension
The prescriber should follow the specific dosing instructions for each formulation. The tablet and oral suspension are not to be used interchangeably due to the differences in the dosing of each formulation.
Posaconazole Biocon delayed release tablets 100 mg
Posaconazole Biocon delayed release tablets 100 mg may be taken without regard to food intake.
Posaconazole Biocon delayed release tablets 100 mg should be swallowed whole, and not be divided, crushed, or chewed.
Use in renal impairment: No dose adjustment is required for renal dysfunction and Posaconazole Biocon delayed release tablets 100 mg is not significantly renally eliminated, an effect of severe renal insufficiency on the pharmacokinetics of Posaconazole Biocon delayed release tablets 100 mg is not expected and no dose adjustment is recommended.
Use in hepatic impairment: There are limited pharmacokinetic data in patients with hepatic insufficiency; therefore, no recommendation for dose adjustment can be made. In the small number of subjects studied who had hepatic insufficiency, there was an increase in half-life with a decrease in hepatic function.
Use in children: Safety and efficacy in children below the age of 13 years have not been established.
Contraindications
4.3 Contraindications
Posaconazole Biocon delayed release tablets 100 mg is contraindicated in patients with known hypersensitivity to Posaconazole Biocon delayed release tablets 100 mg or any component of the product.
Although not studied in vitro or in vivo, coadministration of the CYP3A4 substrates terfenadine, astemizole, cisapride, pimozide, or quinidine with Posaconazole Biocon delayed release tablets 100 mg are contraindicated since increased plasma concentrations of these drugs can lead to QT prolongation and rare occurrences of torsade de pointes.
Coadministration with the HMG-CoA reductase inhibitors that are primarily metabolized through CYP3A4 is contraindicated since increased plasma concentration of these drugs can lead to rhabdomyolysis.
Although not studied in vitro or in vivo, Posaconazole Biocon delayed release tablets 100 mg may increase the plasma concentrations of ergot alkaloids which may lead to ergotism. Coadministration of Posaconazole Biocon delayed release tablets 100 mg and ergot alkaloids is contraindicated.
