LAZCLUZE FILM-COATED TABLETS 240MG [SIN17348P]
Active ingredients: LAZCLUZE FILM-COATED TABLETS 240MG
Product Info
LAZCLUZE FILM-COATED TABLETS 240MG
[SIN17348P]
Product information
Active Ingredient and Strength | LAZERTINIB MESYLATE MONOHYDRATE EQV LAZERTINIB - 240 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | JANSSEN CILAG SPA - ITALY |
Registration Number | SIN17348P |
Licence Holder | JOHNSON & JOHNSON INTERNATIONAL (SINGAPORE) PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01EB09 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
LAZCLUZE® in combination with amivantamab is indicated for the first-line treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 19 deletions or exon 21 L858R substitution mutations.
Dosing
Dosage and Administration
Dosage (≥ 18 years)
The recommended dosage of LAZCLUZE® is 240 mg orally once daily in combination with amivantamab until disease progression or no longer tolerated by the patient.
It is recommended to administer LAZCLUZE® any time prior to amivantamab when given on the same day. Refer to the amivantamab prescribing information for recommended amivantamab dosing information.
Missed dose(s)
If a dose of LAZCLUZE® is missed, it can be administered within 12 hours. If more than 12 hours have passed since the dose was to be given, do not administer the missed dose and administer the next dose per the usual dosing schedule.
Dose modifications
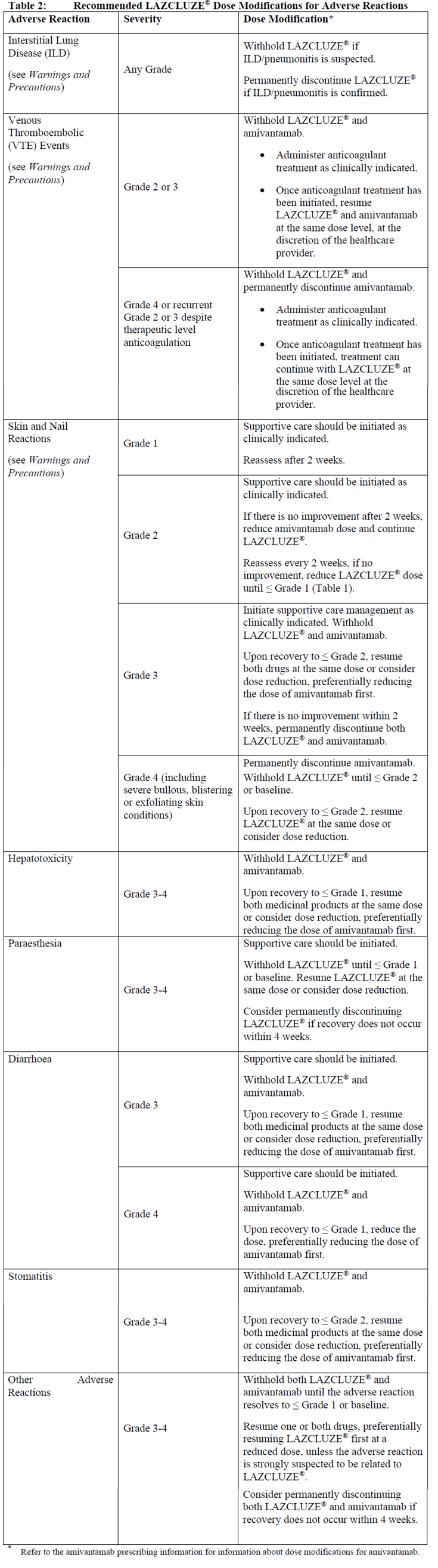
The recommended dose reductions for adverse reactions are presented in Table 1.

Dose modifications for specific adverse reactions are presented in Table 2.
Refer to the amivantamab prescribing information for information about dose modifications for amivantamab.
Special populations
Pediatrics (17 years of age and younger)
The safety and efficacy of LAZCLUZE® have not been established in pediatric patients.
Elderly (65 years of age and older)
Among 421 patients with non-small cell lung cancer treated with LAZCLUZE® in combination with amivantamab in NSC3003, 44.7% were 65 years and older and 11.6% were 75 years and older.
No overall differences in safety or effectiveness were observed as a function of age. No dose adjustment based on age is required (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No formal studies of lazertinib in patients with renal impairment have been conducted.
No dose adjustment is required for patients with mild or moderate renal impairment. LAZCLUZE® has not been studied in patients with severe renal impairment or end-stage renal disease (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No dose adjustment is required for patients with mild or moderate hepatic impairment. The PK of lazertinib in patients with severe hepatic impairment is unknown (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administration
This medicinal product is for oral use. Swallow tablets whole with or without food. Do not crush, split, or chew the tablet.
If vomiting occurs any time after taking LAZCLUZE®, take the next dose the next day.
Contraindications
Contraindications
Hypersensitivity to the active substance(s) or to any of the excipients.
