ELAHERE CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/20ML [SIN17405P]
Active ingredients: ELAHERE CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/20ML
Product Info
ELAHERE CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/20ML
[SIN17405P]
Product information
Active Ingredient and Strength | MIRVETUXIMAB SORAVTANSINE - 100 MG/20 ML |
Dosage Form | INFUSION, SOLUTION CONCENTRATE |
Manufacturer and Country | BSP PHARMACEUTICALS S.P.A. - ITALY |
Registration Number | SIN17405P |
Licence Holder | ABBVIE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FX26 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
ELAHERE as monotherapy is indicated for the treatment of adult patients with folate receptor-alpha (FRα) positive, platinum-resistant high grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens (see section 4.2).
Dosing
4.2 Posology and method of administration
ELAHERE must be initiated and supervised by a physician experienced in the use of anticancer medicinal products.
Patient selection
Eligible patients should have FRα tumour status defined as ≥75% viable tumour cells demonstrating moderate (2+) and/or strong (3+) membrane staining by immunohistochemistry (IHC), assessed by a CE-marked in vitro diagnostic (IVD) with the corresponding intended purpose. If a CE-marked IVD is not available, an alternative validated test should be used.
Posology
The recommended dose of ELAHERE is 6 mg/kg adjusted ideal body weight (AIBW) administered once every 3 weeks (21-day cycle) as an intravenous infusion until disease progression or unacceptable toxicity. Dosing based on AIBW reduces exposure variability for patients who are either underweight or overweight.
The total dose of ELAHERE is calculated based on each patient’s AIBW using the following formula:

Pre-medication
Pre-medication for infusion related reactions (IRRs), nausea, and vomiting
Administer the pre-medications in Table 1 prior to each infusion of ELAHERE to reduce the incidence and severity of IRRs, nausea, and vomiting.

For patients experiencing nausea and/or vomiting, additional antiemetics may be considered thereafter as needed.
For patients who experience an IRR Grade ≥2, additional pre-medication with dexamethasone 8 mg two times a day (BID) (or equivalent) the day before ELAHERE administration should be considered.
Ophthalmic exam and pre-medication
Ophthalmic exam: An ophthalmic exam including visual acuity and slit lamp exam should be conducted before the initiation of ELAHERE and if a patient develops any new or worsening ocular symptoms prior to the next dose. In patients with ≥ Grade 2 ocular adverse reactions, additional ophthalmic exams should be conducted at a minimum of every other cycle and as clinically indicated until resolution or return to baseline.
Ophthalmic topical steroids: For patients found to have signs of ≥ Grade 2 corneal adverse reactions (keratopathy) on slit lamp examination, secondary prophylaxis with ophthalmic topical steroids is recommended for subsequent cycles of ELAHERE, unless the patient’s eye care professional determines that the risks outweigh the benefits of such therapy.
Patients should be instructed to use steroid eye drops on the day of infusion and through the next 7 days of each subsequent cycle of ELAHERE (see Table 3).
Patients should be advised to wait at least 15 minutes after ophthalmic topical steroid administration before instilling lubricating eye drops.
During treatment with ophthalmic topical steroids the measurement of intraocular pressure and an examination with slit lamp should be carried out regularly.
Lubricating eye drops: It is recommended to instruct patients to use lubricating eye drops throughout treatment with ELAHERE.
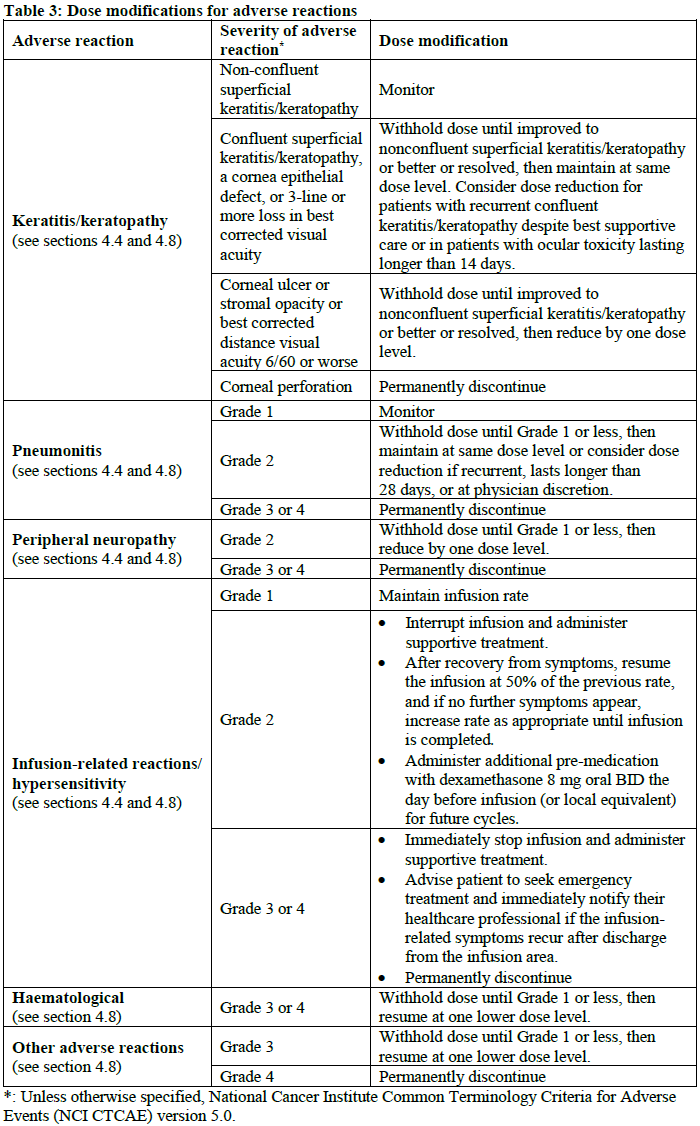
Dose modifications
Before the start of each cycle, the patient should be advised to report any new or worsening symptoms to the treating physician or qualified individual.
In patients who develop new or worsening ocular symptoms, an ophthalmic exam should be conducted before dosing. The treating physician should review the patient’s ophthalmic examination report before dosing and determine the dose of ELAHERE based on the severity of findings in the most severely affected eye.
Table 2 and Table 3 provide dose reductions and modifications for adverse reactions. The schedule of administration should be maintained at a 3-week interval between the doses.

Special populations
Paediatric population
There is no relevant use of ELAHERE for the treatment of epithelial ovarian, fallopian tube, or primary peritoneal cancer in the paediatric population.
Elderly
No dose adjustment of ELAHERE is recommended in patients ≥65 years of age (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No dose adjustment of ELAHERE is recommended for patients with mild to moderate renal impairment (creatinine clearance [CLcr] 30 to <90 mL/min). ELAHERE has not been evaluated in patients with severe renal impairment (CLcr 15 to <30 mL/min) or end-stage renal disease and the potential need for dose adjustment in these patients cannot be determined (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No dose adjustment of ELAHERE is recommended for patients with mild hepatic impairment (total bilirubin ≤ upper limit of normal [ULN] and aspartate aminotransferase [AST] > ULN or total bilirubin >1 to 1.5 times ULN and any AST) (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
ELAHERE should be avoided in patients with moderate to severe hepatic impairment (total bilirubin >1.5 ULN with any AST).
Method of administration
ELAHERE is for intravenous infusion at a rate of 1 mg/min. If well tolerated after 30 minutes, the infusion rate can be increased to 3 mg/min. If well tolerated after 30 minutes at 3 mg/min, the infusion rate can be increased to 5 mg/min.
For incompatibilities, see section 6.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
ELAHERE requires dilution with 5% glucose for intravenous infusion. For instructions on dilution of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
ELAHERE must be administered as an intravenous infusion only, using a 0.2 or 0.22 micrometre polyethersulfone (PES) in-line filter (see Special handling and disposal procedures in section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Precautions to be taken before handling or administering the medicinal product
This medicinal product contains a cytotoxic component, which is covalently attached to the monoclonal antibody (see special handling and disposal procedures in section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
