EMBLAVEO POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 1.5G/0.5G [SIN17438P]
Active ingredients: EMBLAVEO POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 1.5G/0.5G
Product Info
EMBLAVEO POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 1.5G/0.5G
[SIN17438P]
Product information
Active Ingredient and Strength | AVIBACTAM SODIUM EQV AVIBACTAM - 0.5 G/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | HOSPIRA AUSTRALIA PTY LTD - AUSTRALIA |
Registration Number | SIN17438P |
Licence Holder | PFIZER PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01DF51 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
EMBLAVEO is indicated for the treatment of the following infections in adult patients (see sections 4.4 and 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information):
Complicated intra-abdominal infection (cIAI)
Hospital-acquired pneumonia (HAP), including ventilator-associated pneumonia (VAP)
Complicated urinary tract infection (cUTI), including pyelonephritis
Consideration should be given to official guidance on the appropriate use of antibacterial agents.
Dosing
4.2 Posology and method of administration
Posology
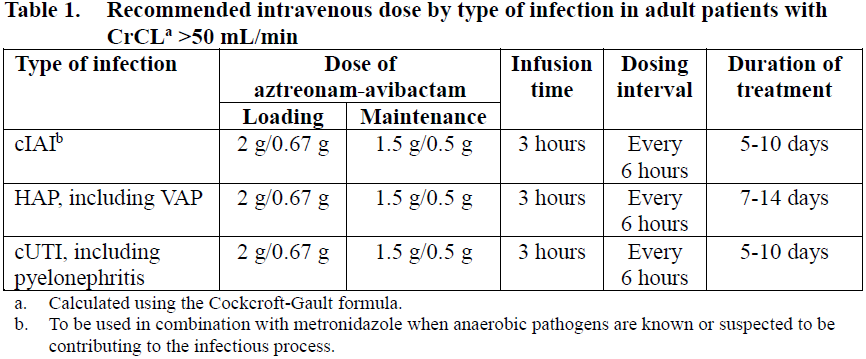
Dose in adults with estimated creatinine clearance (CrCL) >50 mL/min
Table 1 shows the recommended intravenous dose for patients with a creatinine clearance (CrCL) >50 mL/min. A single loading dose is followed by maintenance doses beginning at the next dosing interval.
Special populations
Elderly
No dosage adjustment is required in elderly patients based on age (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
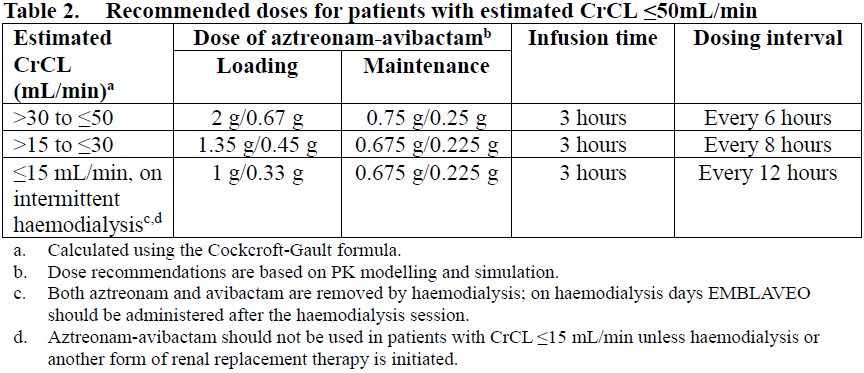
No dosage adjustment is required in patients with mild renal impairment (estimated CrCL >50 to ≤80 mL/min).
Table 2 shows the recommended dose adjustments for patients with estimated creatinine clearance ≤50 mL/min. A single loading dose is followed by maintenance doses beginning at the next dosing interval.
In patients with renal impairment, close monitoring of estimated creatinine clearance is advised (see sections 4.4 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
There are insufficient data to make dosing adjustment recommendations for patients undergoing renal replacement therapy other than haemodialysis (e.g., continuous veno-venous hemofiltration or peritoneal dialysis). Patients receiving continuous renal replacement therapy (CRRT) need a higher dose than patients on haemodialysis. For patients receiving continuous renal replacement therapy, the dose should be adjusted guided by the CRRT clearance (CLCRRT in mL/min).
Hepatic impairment
No dosage adjustment is required in patients with hepatic impairment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
The safety and efficacy of EMBLAVEO in paediatric patients <18 years of age have not yet been established. No data are available.
Method of administration
Intravenous use.
EMBLAVEO is administered by intravenous infusion over 3 hours.
For instructions on reconstitution and dilution of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Severe hypersensitivity (e.g., anaphylactic reaction, severe skin reaction) to any other type of beta-lactam antibacterial agent (e.g., penicillins, cephalosporins or carbapenems).
